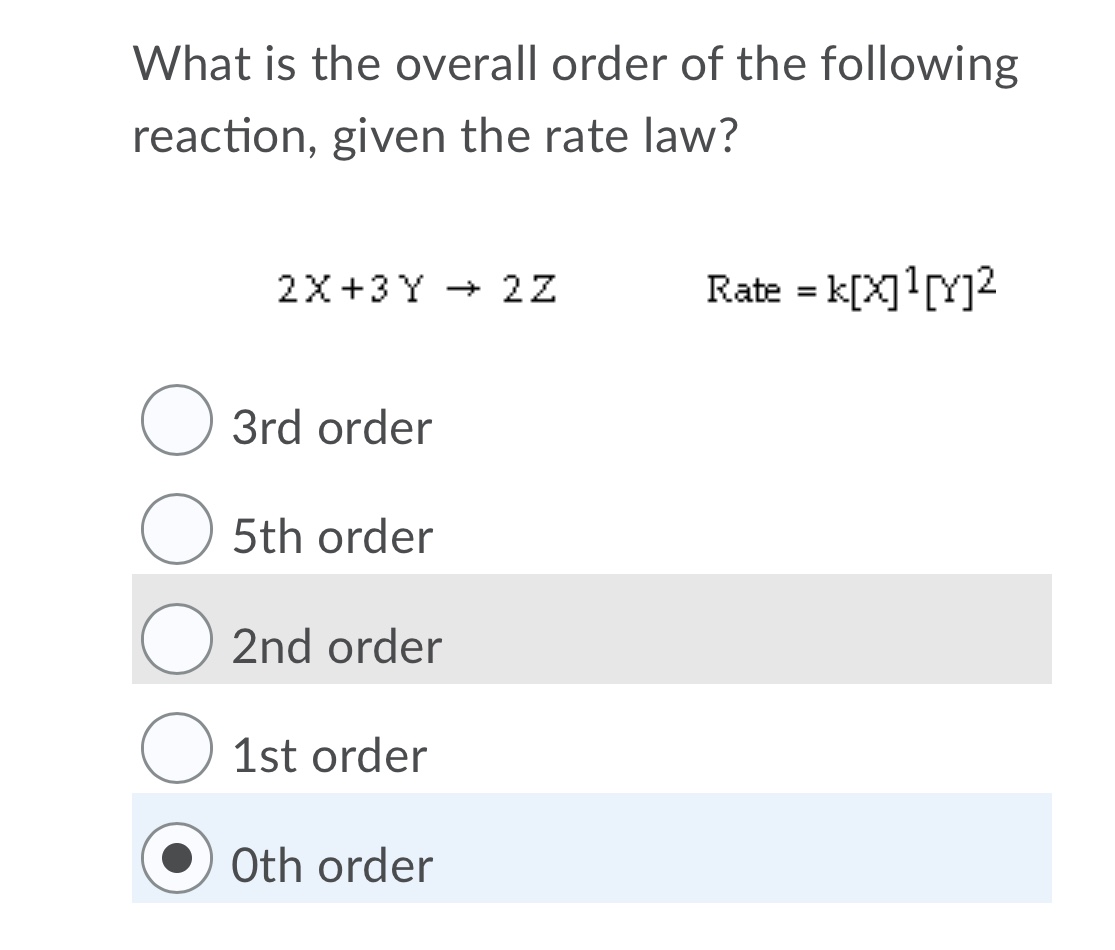
How to Determine the Order of Reaction by Comparing Initial Rates of Reactions | Chemistry | Study.com
![SOLVED: What is the overall reaction order for the reaction that has the rate law Rate = k[O2][NO]^2? a) zero order b) first order c) second order d) third order SOLVED: What is the overall reaction order for the reaction that has the rate law Rate = k[O2][NO]^2? a) zero order b) first order c) second order d) third order](https://cdn.numerade.com/ask_previews/bea16924-e043-4592-9ba1-7c64b3c6ae29_large.jpg)
SOLVED: What is the overall reaction order for the reaction that has the rate law Rate = k[O2][NO]^2? a) zero order b) first order c) second order d) third order
![The following results have been obtained during the kinetic studies of the reaction: 2A + B → C + D Experiment [A]/mol L^-1 [B]/mol L^-1 Initial rate of formation of D/mol L^-1 The following results have been obtained during the kinetic studies of the reaction: 2A + B → C + D Experiment [A]/mol L^-1 [B]/mol L^-1 Initial rate of formation of D/mol L^-1](https://dwes9vv9u0550.cloudfront.net/images/8845933/08b4ef5f-addd-47bf-9960-e6214462471c.jpg)

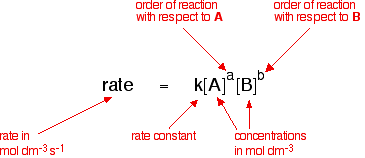
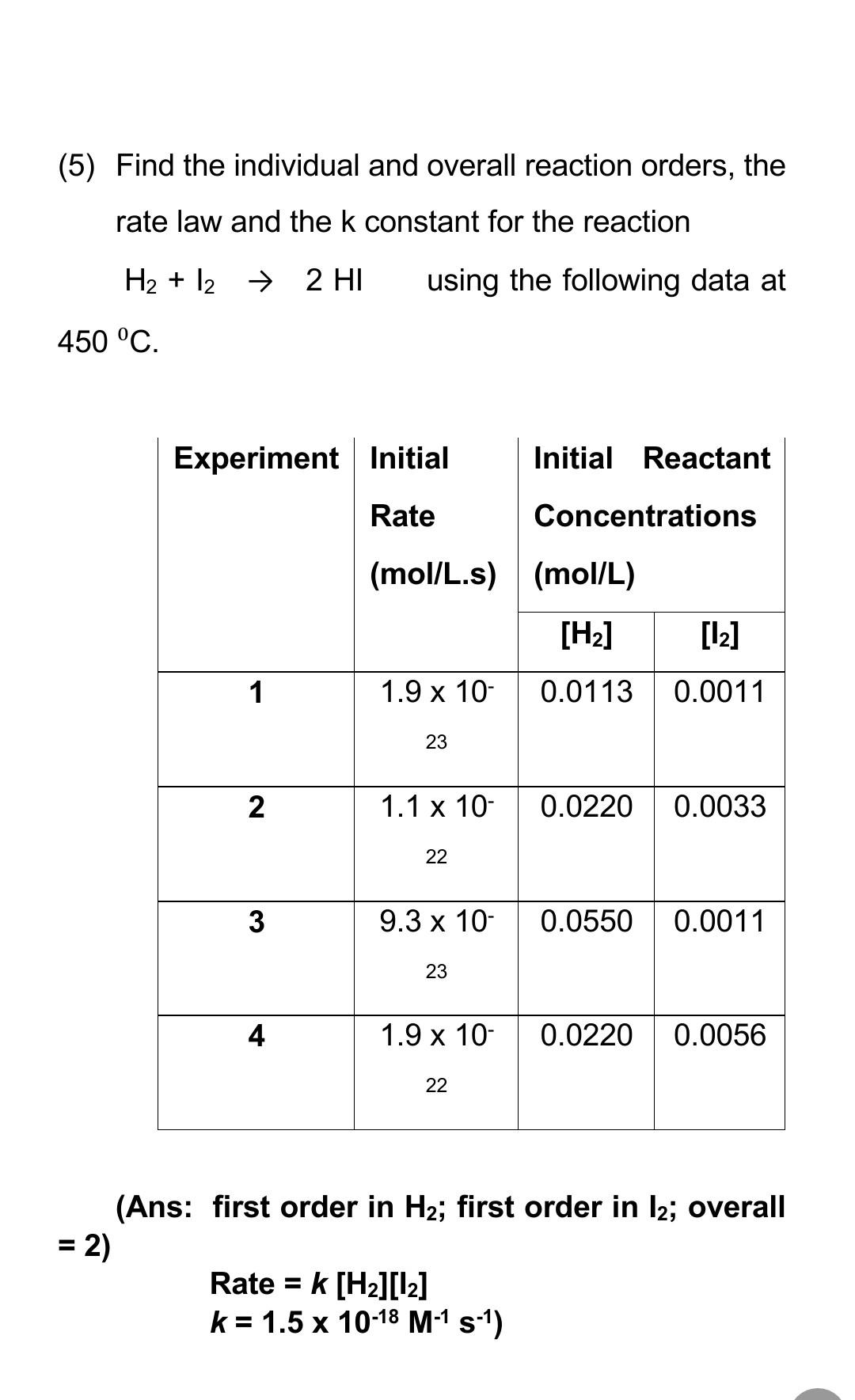

![16.1/R2.2.6 Rate constant, overall order of reaction, order of reaction [HL IB Chemistry] - YouTube 16.1/R2.2.6 Rate constant, overall order of reaction, order of reaction [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/9sMFJMuZzmg/maxresdefault.jpg)