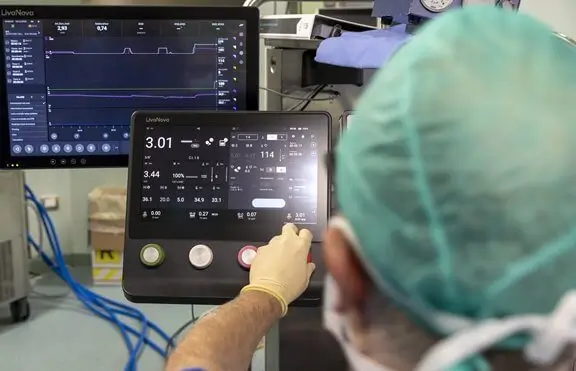
LivaNova Initiates Limited Commercial Release in Europe of the Essenz Perfusion System for Cardiopulmonary Bypass Procedures | Business Wire

LivaNova Receives U.S. FDA 510(k) Clearance for Essenz Heart-Lung Machine for Cardiopulmonary Bypass Procedures | Business Wire

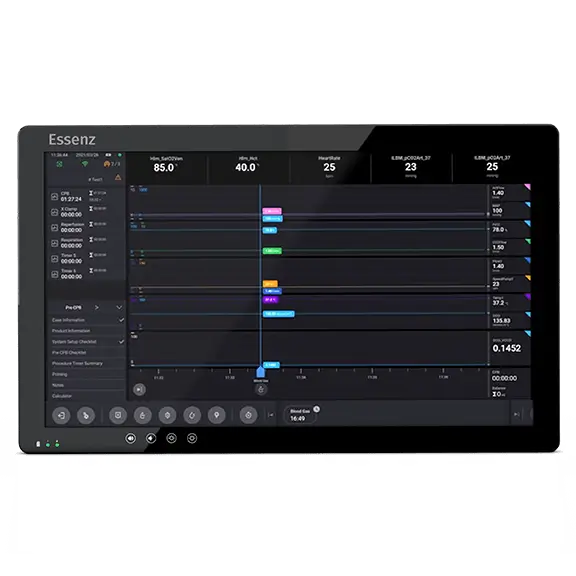
LivaNova Initiates Targeted Commercial Launch of the Essenz Patient Monitor for Cardiopulmonary Bypass Procedures | Business Wire