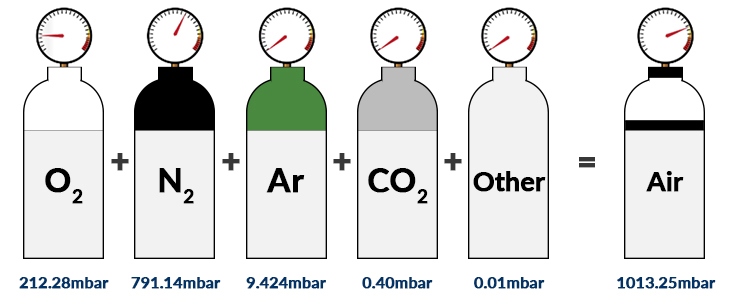
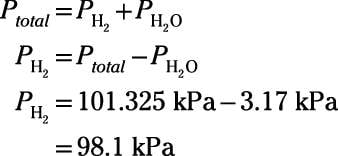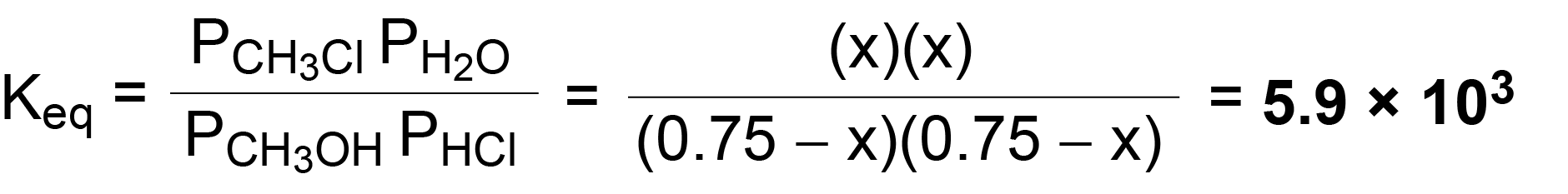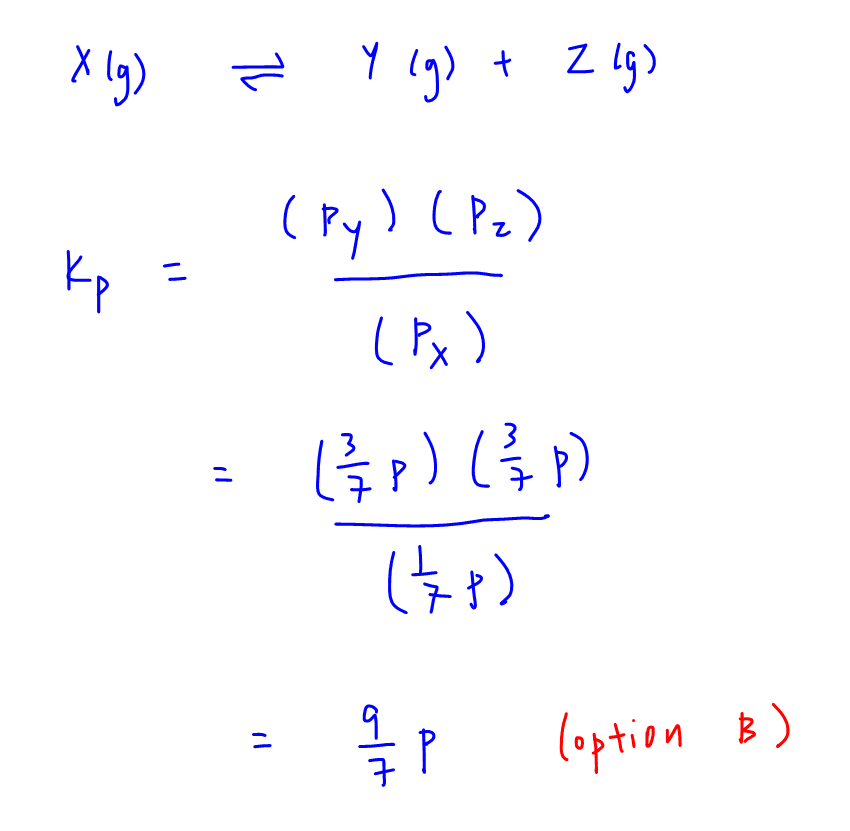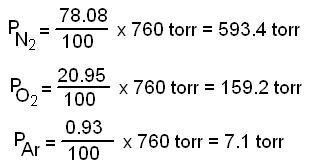SOLVED: Calculate the partial pressure of CO2 (in mmHg) collected over water in an experiment, if the temperature was 19.2 ºC and the barometric pressure was 708.2 mmHg. Refer to the Table

Question Video: Calculating the Equilibrium Constant for Partial Pressures Given the Partial Pressure of Each Species | Nagwa

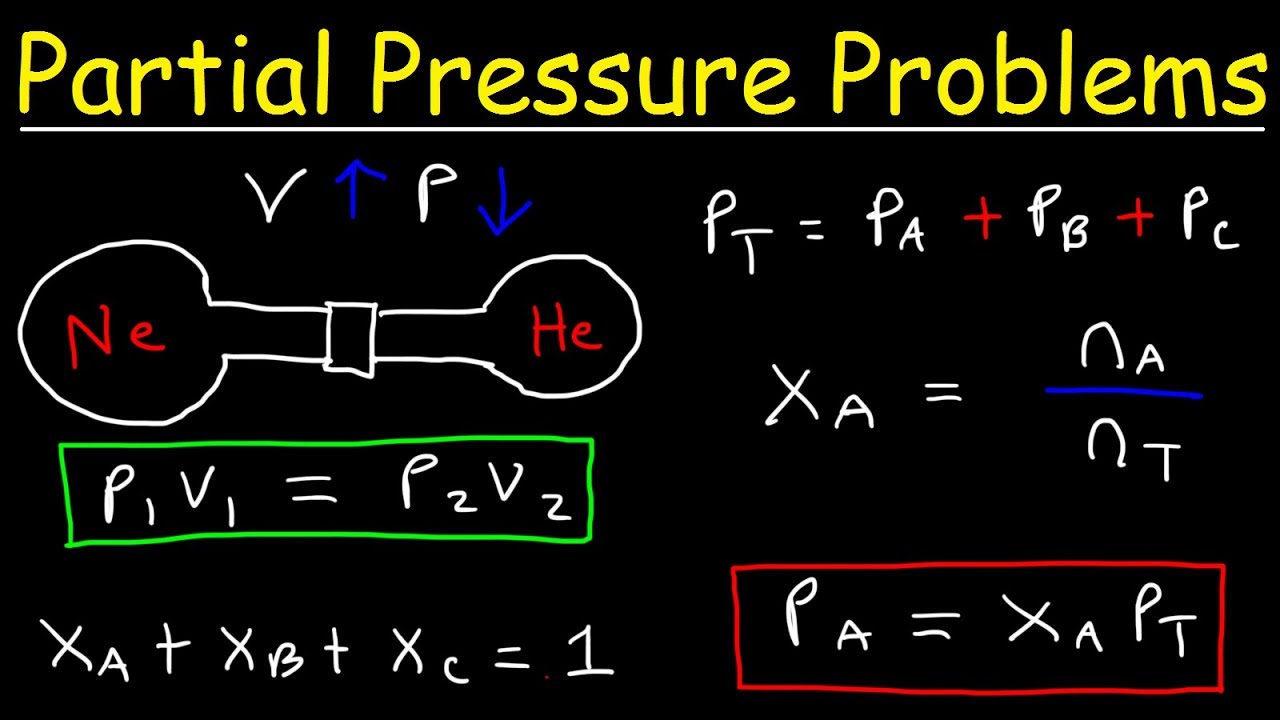
I don't understand the calculations behind Partial Pressure in this case. How can you just subtract the partial pressures as if it is concentrations to find the partial pressures at equilibrium. 6p-1/7p