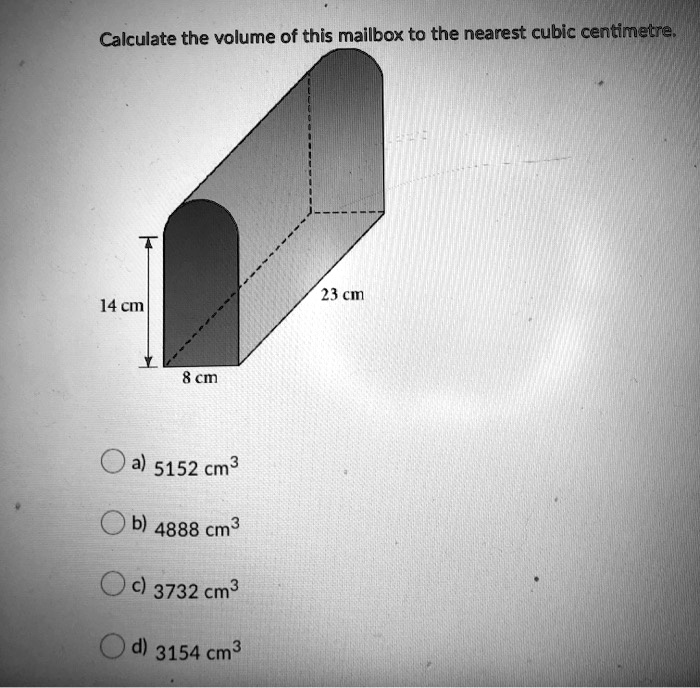
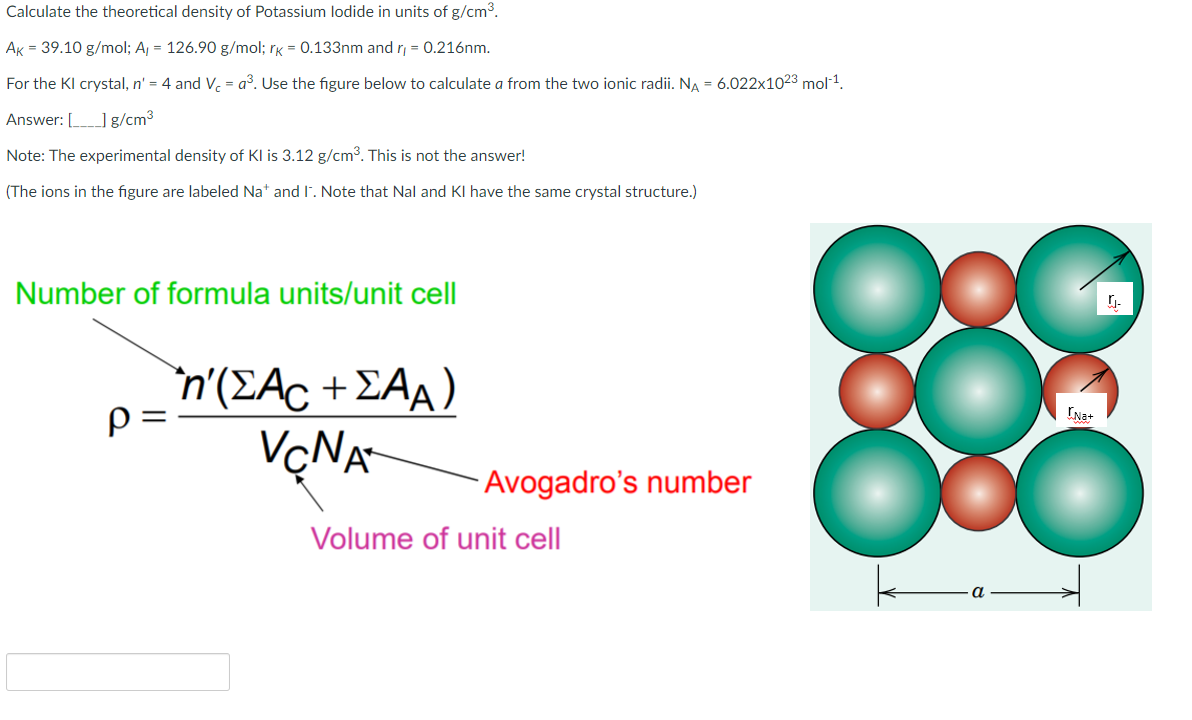
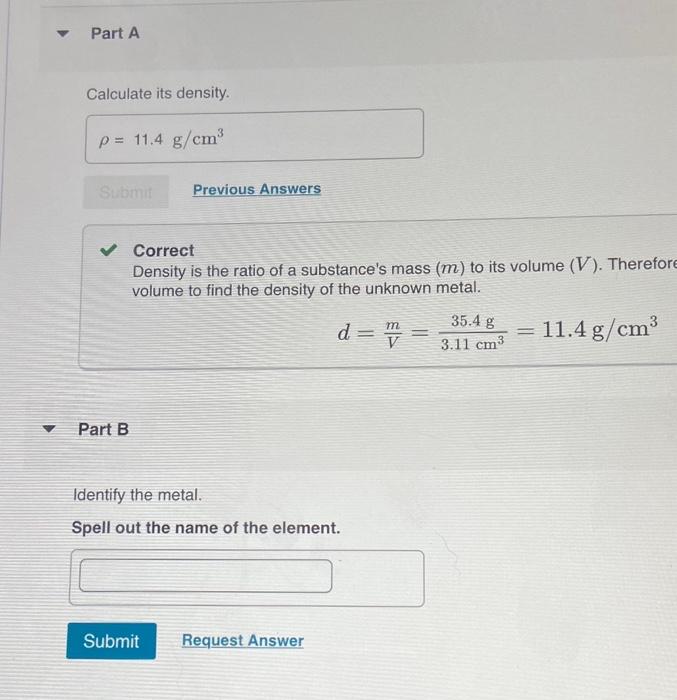
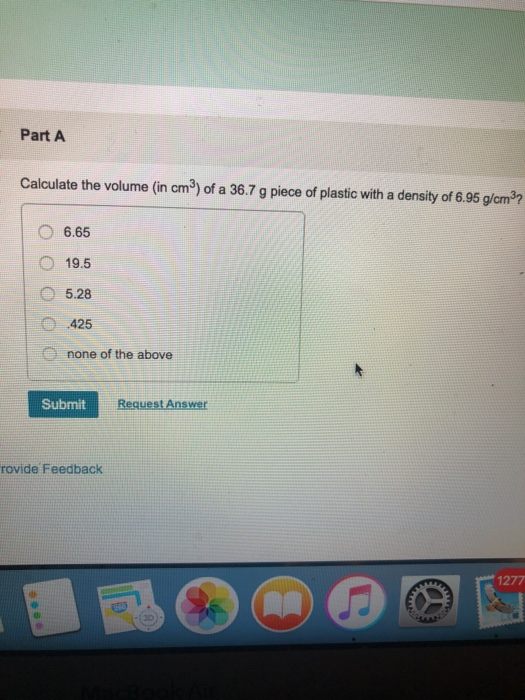
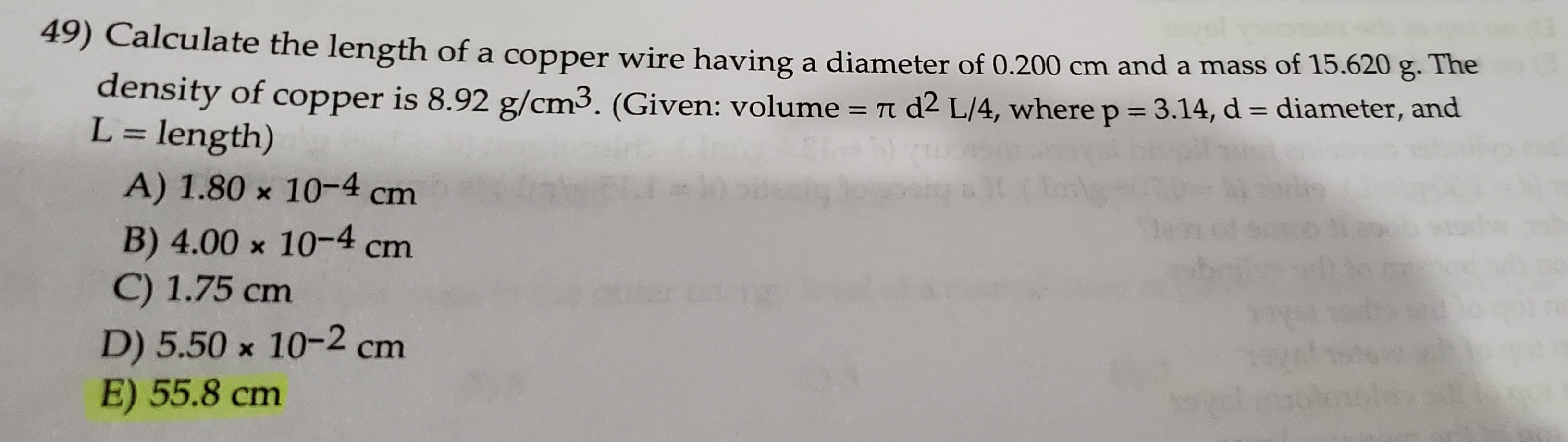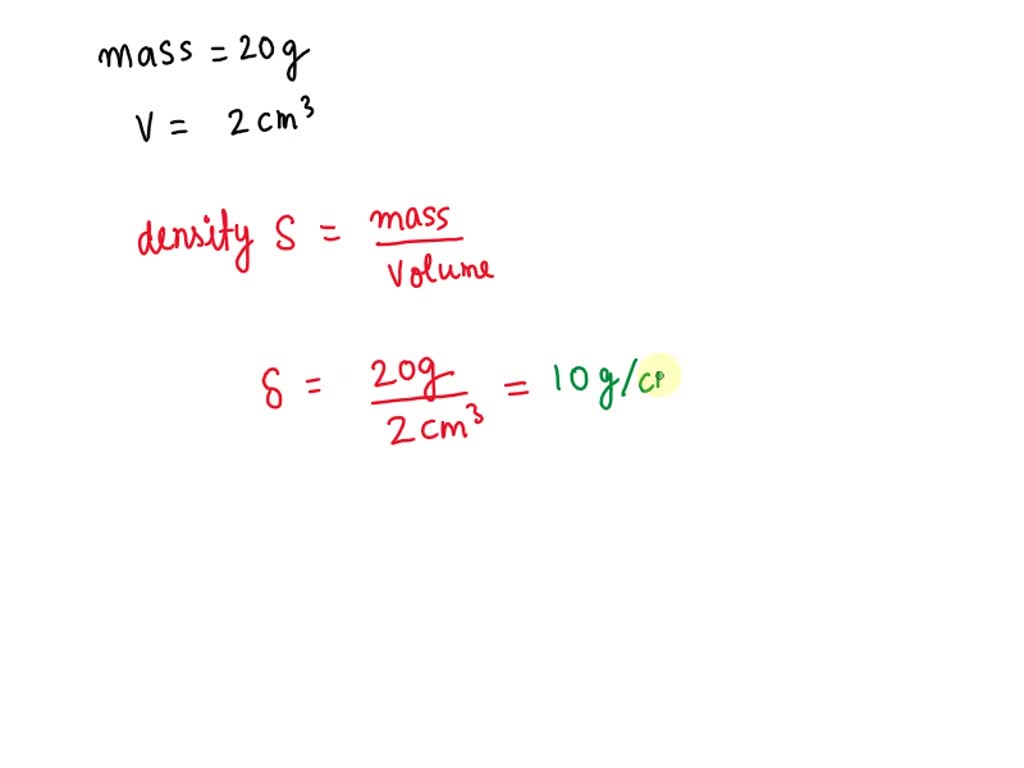SOLVED: For the masses and volumes indicated, calculate the density in grams per cubic centimeter. (a) mass = 454.1 g; volume = 291 cm3 =g/cm3 (b) mass = 0.38 lb; volume =
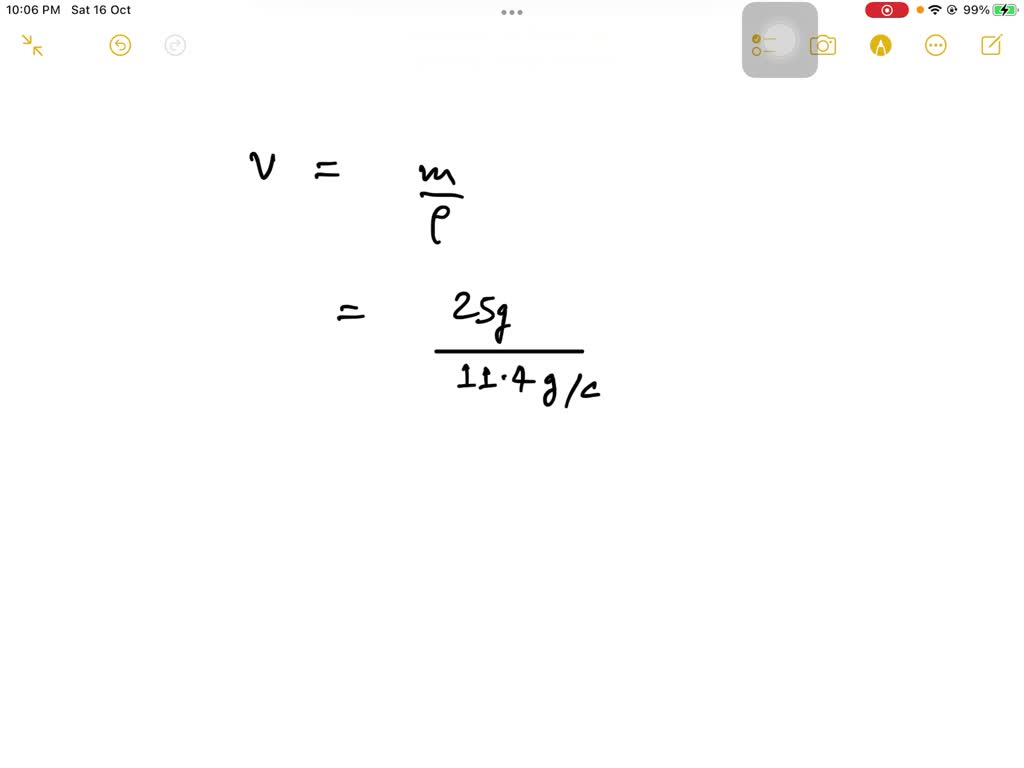
SOLVED: The density of lead is 11.4 g/cm 3 at 25*C. Calculate the volume occupied by 25.0 g of lead. 0.456 cm3 0 2.19 cm3 285 cm3 1.24 cm3 6.05 cm3
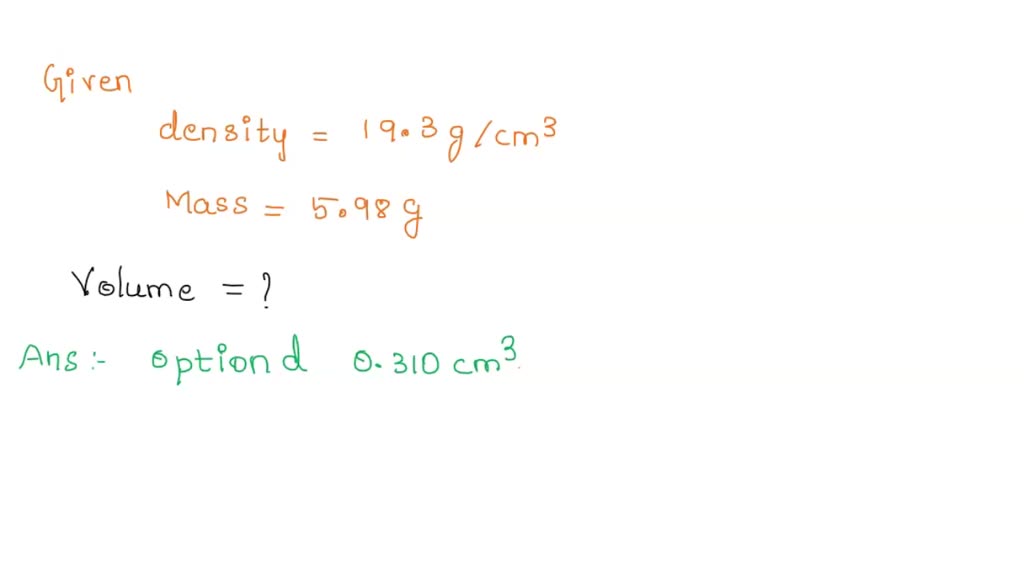
SOLVED: Given that the density of gold is 19.3 g/cm3 , calculate the volume (in cm3 ) of a gold nugget with a mass of 5.98 g. a) 3.23 cm3 d) 0.310